Hvidberg Compliance Management
Smart, streamlined Clinical Development governance
— fit-for-purpose today, scalable for tomorrow
Turning Clinical Development Friction into Clarity and Compliance
- At Hvidberg Compliance Management (HCM), we support biotech, medical device and pharmaceutical companies by strengthening operating principles and oversight, streamlining processes, and resolving cross‑functional clinical development inefficiencies.
Complexity to Clarity & Compliance ™ Model
Our framework connects governance, operations and compliance and builds clarity into how work gets done.
Built around three phases – Define, Develop, Deploy – the model aligns
- Regulations
- People
- Operations
We ensure every strategy is practical, compliant, and scalable by working through the phases:
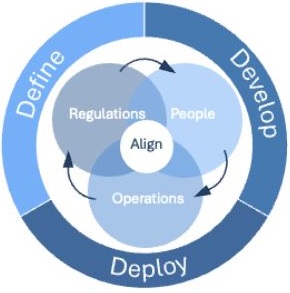
- Define: Diagnose root causes across the clinical development ecosystem — governance, processes, tools, roles and cross‑functional collaboration.
- Develop: Co‑design governance structures, operating principles and process‑ & tool‑aligned ways of working that fit organisational maturity.
- Deploy: Embed operating principles through training, oversight and clear ownership to ensure sustainable adoption.
This approach replaces operational complexity with clarity, strengthens oversight and compliance, and streamlines processes and cross‑functional collaboration across clinical development.
Why Choose Hvidberg Compliance Management?
Clinical development is more complex than ever. Sponsors must demonstrate robust risk management, transparent oversight, and efficient delivery
– often across outsourced partnerships. HCM guide life science organisations navigate this complexity with clarity, compliance, and control.
What makes HCM different?
Deep Experience
Bringing structure and clarity by aligning people, processes and digital solutions across clinical developmen.
Trusted Oversight
Frameworks aligned with ICH/GCP requirements for full compliance and transparency.
Practical Solutions
Processes designed for efficiency, compliance, and long-term impact.
Collaborative Approach
Strong partnerships that align stakeholders and build high-performing teams.
Resilience & Integrity
Recognised for professionalism, empathy, and foresight in guiding clients through change.
Our mission: To enable you to deliver compliant, efficient and impactful development — today and in the future.
SERVICES
Every engagement is guided by our Complexity to Clarity and Compliance™ Model, ensuring strategies are compliant, feasible, and fully embedded in your organisation.
Our three-step approach:
- Define the challenge with precision.
- Root cause analysis based on current practices and stakeholder input.
- Clear scope definition.
- Resolution scenarios with time and effort assessments.
- Develop tailored, compliant solutions.
- Detailed deliverable plan based on agreed scenario.
- Start-up, execution, status reporting, and implementation plan.
- Anchoring and handover strategy.
- Debrief and lessons learned.
- Deploy the strategy with training, oversight, and anchoring.
- Execute implementation plan with process updates, training, and hyper-care.
- Deliver anchoring, handover and ownership plan.
- Debrief and lessons learned.
Service Customisations
Our services are flexible and can be tailored to your needs:
Full Phase or Phase Segment Delivery
- HCM can deliver entire phases or specific segments.
- Example: managing deliverable planning in the Development Phase while your team creates core content.
Full or Phased Engagement
- HCM can deliver all phases from start to finish.
- Or targeted support, for example training during the Deploy Phase.
Fit-for-purpose tools and documentation
- Always aligned with customer standards.
- Tailored tools provided when needed.

Clinical Operations Strategies
Oversight frameworks for sponsor quality, compliance, and study progress

Complex Problem Resolution
Untangle infrastructure issues, provide short-term fixes and long-term change scenarios.

Change & Transition Management
Communications, training, and oversight to ensure lasting adoption.

